Atmosphere
Layers
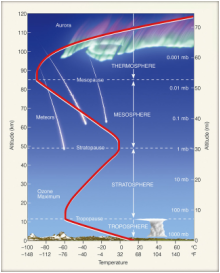
The atmosphere is an envelope of gases that surrounds the planet. As with just about anything we'll study, the atmosphere has layers. These layers are determined or divided by changes in temperature trends. In each layer, the temperature either increases or decreases with altitude. Between each layer, the temperature stops changing and will go the other direction in the next layer. This is called a pause.
Exosphere - Beginning of space
Magnetosphere - Magnetic field is created by metal outer core moving over metal inner core; protects the Earth from cosmic radiation and solar wind
Ionosphere - Sun's radiation splits molecules apart; radio signals bounce off of the ions to come back to the surface
Thermosphere - Air is so thin, we need lasers to measure its temperature, but it goes up
Mesopause - Change
Mesosphere - Temperature goes back down
Stratopause - Change
Stratosphere - Ozone layer moves up and down in this layer; temperature goes up
Tropopause - Change
Troposphere - Layer we live in; all of the weather happens in this layer; temperature decreases with altitude
The ozone layer is a thin layer of a special form of oxygen. Normally, oxygen exists as two oxygen atoms bonded together. But ozone is three oxygen atoms bonded together. It's a toxic gas if we breathe it, but up in the atmosphere it absorbs ultraviolet (UV) rays from the Sun. UV rays would destroy DNA and prevent life from thriving on the surface. When it absorbs UV radiation, the temperature of the surrounding air heats up. This is why the stratosphere heats up.
The atmosphere is an envelope of gases that surrounds the planet. As with just about anything we'll study, the atmosphere has layers. These layers are determined or divided by changes in temperature trends. In each layer, the temperature either increases or decreases with altitude. Between each layer, the temperature stops changing and will go the other direction in the next layer. This is called a pause.
Exosphere - Beginning of space
Magnetosphere - Magnetic field is created by metal outer core moving over metal inner core; protects the Earth from cosmic radiation and solar wind
Ionosphere - Sun's radiation splits molecules apart; radio signals bounce off of the ions to come back to the surface
Thermosphere - Air is so thin, we need lasers to measure its temperature, but it goes up
Mesopause - Change
Mesosphere - Temperature goes back down
Stratopause - Change
Stratosphere - Ozone layer moves up and down in this layer; temperature goes up
Tropopause - Change
Troposphere - Layer we live in; all of the weather happens in this layer; temperature decreases with altitude
The ozone layer is a thin layer of a special form of oxygen. Normally, oxygen exists as two oxygen atoms bonded together. But ozone is three oxygen atoms bonded together. It's a toxic gas if we breathe it, but up in the atmosphere it absorbs ultraviolet (UV) rays from the Sun. UV rays would destroy DNA and prevent life from thriving on the surface. When it absorbs UV radiation, the temperature of the surrounding air heats up. This is why the stratosphere heats up.
Composition
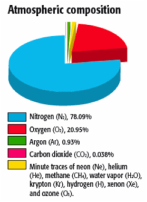
Earth's atmosphere is significantly different from the other planets' atmospheres. We'll examine each planet's atmosphere when we tour through the Solar System later. Right now, though, we're taking a look at the gases that keep us alive here on Earth.
78% - Nitrogen is the most abundant gas in the atmosphere
21% - Oxygen is the second most abundant gas, but it's the one that keeps us alive.
0.9% - Argon is the third most abundant gas; it's an inert gas (does not react with anything)
less than 0.1% - all of the other gases found: water vapor, carbon dioxide, ammonia, etc.
Oxygen levels have fluctuated throughout Earth's history. Early on, there was no oxygen. About 300 million years ago, oxygen levels went up to as high as 30%. Now, it's 21%.
Earth's atmosphere is significantly different from the other planets' atmospheres. We'll examine each planet's atmosphere when we tour through the Solar System later. Right now, though, we're taking a look at the gases that keep us alive here on Earth.
78% - Nitrogen is the most abundant gas in the atmosphere
21% - Oxygen is the second most abundant gas, but it's the one that keeps us alive.
0.9% - Argon is the third most abundant gas; it's an inert gas (does not react with anything)
less than 0.1% - all of the other gases found: water vapor, carbon dioxide, ammonia, etc.
Oxygen levels have fluctuated throughout Earth's history. Early on, there was no oxygen. About 300 million years ago, oxygen levels went up to as high as 30%. Now, it's 21%.
