Minerals
Characteristics
What makes a mineral a mineral? An object must meet 5 criteria before being called a mineral. These are the mineral's characteristics.
I like to suggest using the ice test: ice is made in nature (1); ice is not alive (2); ice is solid water (3); ice is made of H2O (4); and ice is made of hexagons (5) which is why snowflakes are 6-sided.
What makes a mineral a mineral? An object must meet 5 criteria before being called a mineral. These are the mineral's characteristics.
- Natural: it cannot be man-made or artificial.
- Inorganic: it cannot be organic (this word causes all sorts of confusion because of how nutritionists use it; on food, organic means chemical-free which I guess implies that it's a vacuum as only a vacuum is chemical-free); it cannot be alive or ever have been alive (the true definition of organic).
- Solid: it cannot be a liquid or a gas.
- Chemical composition: it must have a definite chemical formula to describe it.
- Crystal structure: it must have a regularly, repeating structure of atoms
I like to suggest using the ice test: ice is made in nature (1); ice is not alive (2); ice is solid water (3); ice is made of H2O (4); and ice is made of hexagons (5) which is why snowflakes are 6-sided.
Properties
Once we establish that we have a mineral, we need to find out what mineral it is. For this, we'll use the mineral's properties to help us identify it. A property is a trait that we use to classify minerals.

Color
Color is probably the worst property. Many minerals are the same color, but some minerals can be different colors...sometimes in the same crystal. For example, quartz exists as crystal (clear), milky (white), amethyst (purple), smoky (gray/brown), rose (pink), and citrine (yellow). Fluorite comes in purple, blue, green, yellow, pink, and white. Tourmaline exists in a similar palette of colors and the variety known as elbaite is pink and green in the same crystal...it's sometimes called watermelon tourmaline. So, even though it's the first thing we see and some minerals are known for a distinct color (sulfur comes to mind, but it's not exclusive even for its color), color is unreliable as a stand alone property.
Color is probably the worst property. Many minerals are the same color, but some minerals can be different colors...sometimes in the same crystal. For example, quartz exists as crystal (clear), milky (white), amethyst (purple), smoky (gray/brown), rose (pink), and citrine (yellow). Fluorite comes in purple, blue, green, yellow, pink, and white. Tourmaline exists in a similar palette of colors and the variety known as elbaite is pink and green in the same crystal...it's sometimes called watermelon tourmaline. So, even though it's the first thing we see and some minerals are known for a distinct color (sulfur comes to mind, but it's not exclusive even for its color), color is unreliable as a stand alone property.
Crystal Shape
Having a crystal structure is required of all minerals. But what that crystal looks like depends on the atoms inside the structure. Some form cubes (diamond), some form hexagons (ice), some form pyramids (silicates), and the rest form many, many different shapes. In order to fully understand this property, a deeper understanding of chemistry is required. Thus, this property doesn't figure much into our basic identification tools.
Having a crystal structure is required of all minerals. But what that crystal looks like depends on the atoms inside the structure. Some form cubes (diamond), some form hexagons (ice), some form pyramids (silicates), and the rest form many, many different shapes. In order to fully understand this property, a deeper understanding of chemistry is required. Thus, this property doesn't figure much into our basic identification tools.

Streak
Another property that relies on color, but is far more useful, is a mineral's streak. When a mineral is rubbed across a white (or black), unglazed, ceramic tile, it can leave behind a line of powder on the tile. This is the streak and it can help tell us what mineral we are looking at. Some minerals will not have a streak because they are too hard (we'll discuss hardness in a little bit), but others have a very distinctive streak. Hematite is the best example of this property. Hematite can be found in a variety of external colors and shapes: rusty red, gunmetal gray, dark silver, almost black; and shapeless or smooth blobs. But, no matter what color or shape it is, it will always have a red-brown streak. It's remarkable when you take a shiny, dark silver mineral and rub it on a streak plate only to see a red-brown powder left behind.
Another property that relies on color, but is far more useful, is a mineral's streak. When a mineral is rubbed across a white (or black), unglazed, ceramic tile, it can leave behind a line of powder on the tile. This is the streak and it can help tell us what mineral we are looking at. Some minerals will not have a streak because they are too hard (we'll discuss hardness in a little bit), but others have a very distinctive streak. Hematite is the best example of this property. Hematite can be found in a variety of external colors and shapes: rusty red, gunmetal gray, dark silver, almost black; and shapeless or smooth blobs. But, no matter what color or shape it is, it will always have a red-brown streak. It's remarkable when you take a shiny, dark silver mineral and rub it on a streak plate only to see a red-brown powder left behind.
Luster
Some minerals are shiny, most are not. The property related to this is called luster. It's not necessarily how shiny a mineral is, but what kind of shiny it is. Basically, it boils down to this: Does it look like gold or silver? If yes, we consider it to look like a metal and have metallic luster. If no, it is nonmetallic. Nonmetallic luster can be further described as just about anything (pearly, waxy, earthy, dull, brilliant, glassy, transparent, translucent, and on and on and on). But our main focus is the difference between metallic and nonmetallic. So, again, does the mineral look like gold or silver? If you think about it, almost all metals look like either gold or silver in some way or another. If it does, then it's metallic. If not, then it's nonmetallic. It's that simple.
Some minerals are shiny, most are not. The property related to this is called luster. It's not necessarily how shiny a mineral is, but what kind of shiny it is. Basically, it boils down to this: Does it look like gold or silver? If yes, we consider it to look like a metal and have metallic luster. If no, it is nonmetallic. Nonmetallic luster can be further described as just about anything (pearly, waxy, earthy, dull, brilliant, glassy, transparent, translucent, and on and on and on). But our main focus is the difference between metallic and nonmetallic. So, again, does the mineral look like gold or silver? If you think about it, almost all metals look like either gold or silver in some way or another. If it does, then it's metallic. If not, then it's nonmetallic. It's that simple.

Cleavage/Fracture
I put these two properties together, because if it's not one, then it's the other. It happens when a mineral breaks. If it breaks along an uneven surface or a curved surface, then we say it fractures. If it breaks along smooth, even planes, then we say it has cleavage. A mineral with cleavage may have 1, 2, or 3 planes of cleavage. If it's 1 plane, we say it has basal cleavage and it splits into sheets. Mica and graphite exhibit basal cleavage. Feldspar exhibits 2 planes of cleavage which is called prismatic. Halite, galena, and diamonds have 3 planes of cleavage which forms cubes. Quartz does not have cleavage, but rather something called conchoidal fracture. This means that it fractures in smooth, circular scoops. This is a feature shared by flint and obsidian which makes them great for arrowheads.
I put these two properties together, because if it's not one, then it's the other. It happens when a mineral breaks. If it breaks along an uneven surface or a curved surface, then we say it fractures. If it breaks along smooth, even planes, then we say it has cleavage. A mineral with cleavage may have 1, 2, or 3 planes of cleavage. If it's 1 plane, we say it has basal cleavage and it splits into sheets. Mica and graphite exhibit basal cleavage. Feldspar exhibits 2 planes of cleavage which is called prismatic. Halite, galena, and diamonds have 3 planes of cleavage which forms cubes. Quartz does not have cleavage, but rather something called conchoidal fracture. This means that it fractures in smooth, circular scoops. This is a feature shared by flint and obsidian which makes them great for arrowheads.
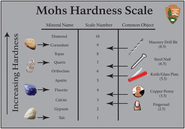
Hardness
This is probably the most subjective property, but is widely used to distinguish minerals from one another. Technically, hardness is an indication of the mineral's ability to resist being scratched. Using a scale of 1-10 (the Mohs Hardness Scale), lower numbers can be more easily scratched and are scratched by higher numbers. Talc is the lowest number at 1. This makes it the softest mineral, getting scratched by anything. Diamond is the highest number at 10. This is the hardest mineral with nothing natural being able to scratch it. Just because it cannot be scratched, though, does not mean it cannot break. Diamonds can break, but not get scratched. So, ladies, if you have a questionable looking diamond, then find a ruby (H=9) and try to scratch the diamond with the ruby. If it is a real diamond, the ruby will do nothing to the diamond. But if it is a fake, it will get scratched.
This is probably the most subjective property, but is widely used to distinguish minerals from one another. Technically, hardness is an indication of the mineral's ability to resist being scratched. Using a scale of 1-10 (the Mohs Hardness Scale), lower numbers can be more easily scratched and are scratched by higher numbers. Talc is the lowest number at 1. This makes it the softest mineral, getting scratched by anything. Diamond is the highest number at 10. This is the hardest mineral with nothing natural being able to scratch it. Just because it cannot be scratched, though, does not mean it cannot break. Diamonds can break, but not get scratched. So, ladies, if you have a questionable looking diamond, then find a ruby (H=9) and try to scratch the diamond with the ruby. If it is a real diamond, the ruby will do nothing to the diamond. But if it is a fake, it will get scratched.

Other Properties
The properties listed above are the most commonly used, but there are a lot of other properties that can be used to easily identify a mineral. For instance, sulfur stinks! It either smells like rotten eggs or gunpowder, depending on your previous personal experiences. Halite is salty...because it is salt. Calcite effervesces (fizzes) in acid. Barite and galena are very dense and, thus, heavy. Fluorite and other minerals, such as apatite, glow or fluoresce under UV light. Uraninite (an ore of uranium) is radioactive. A special variety of calcite called Iceland spar shows something called double refraction. It's virtually clear and when you place it over a picture or words, the light is bent or refracted in such a way as to cause you to see double. And the list can go on. This listing of properties is by no means exhaustive, but highlights some of the more common properties that we come across.
The properties listed above are the most commonly used, but there are a lot of other properties that can be used to easily identify a mineral. For instance, sulfur stinks! It either smells like rotten eggs or gunpowder, depending on your previous personal experiences. Halite is salty...because it is salt. Calcite effervesces (fizzes) in acid. Barite and galena are very dense and, thus, heavy. Fluorite and other minerals, such as apatite, glow or fluoresce under UV light. Uraninite (an ore of uranium) is radioactive. A special variety of calcite called Iceland spar shows something called double refraction. It's virtually clear and when you place it over a picture or words, the light is bent or refracted in such a way as to cause you to see double. And the list can go on. This listing of properties is by no means exhaustive, but highlights some of the more common properties that we come across.
Common Minerals
Rock-forming Minerals
The most common minerals are silicates, which means that they contain silica (SiO2). Oxygen is the most abundant element in the Earth's crust. Silicon is second. When they combine, they make up the silicate family of minerals. These include feldspar (the most abundant mineral on the planet), quartz, micas, and many others. When rocks form, there is a very good chance that they will contain feldspar, quartz, and/or mica. Calcite is also considered a rock-forming mineral because it is the predominant mineral in limestone. But calcite is a carbonate (CO3) and does not belong to the silicate family.
Ore Minerals
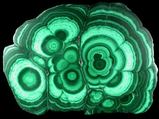
Some minerals are important to us because they contain amounts of metals or other things that we can use. These are the ore minerals. An ore is a rock or mineral containing amounts of a needed substance that we can economically (doesn't cost too much) remove and use. Such minerals include: hematite, pyrite, and magnetite as ores of iron; galena as an ore for lead (used in batteries); graphite as an ore for carbon or just as graphite (pencil lead); and sulfur (used in medicines and gunpowder). Malachite (pictured left) is an ore for copper and bauxite is an ore for aluminum.
The most common minerals are silicates, which means that they contain silica (SiO2). Oxygen is the most abundant element in the Earth's crust. Silicon is second. When they combine, they make up the silicate family of minerals. These include feldspar (the most abundant mineral on the planet), quartz, micas, and many others. When rocks form, there is a very good chance that they will contain feldspar, quartz, and/or mica. Calcite is also considered a rock-forming mineral because it is the predominant mineral in limestone. But calcite is a carbonate (CO3) and does not belong to the silicate family.
Ore Minerals
Some minerals are important to us because they contain amounts of metals or other things that we can use. These are the ore minerals. An ore is a rock or mineral containing amounts of a needed substance that we can economically (doesn't cost too much) remove and use. Such minerals include: hematite, pyrite, and magnetite as ores of iron; galena as an ore for lead (used in batteries); graphite as an ore for carbon or just as graphite (pencil lead); and sulfur (used in medicines and gunpowder). Malachite (pictured left) is an ore for copper and bauxite is an ore for aluminum.
